By Mary K. Coyle and Frank M. Wilhelm, Department of Fish and Wildlife Sciences, College of Natural Resources, University of Idaho
Resembling masses of wet toilet paper, nuisance mats of Didymosphenia geminata – a microscopic attached diatom (Figure 1A) – can smother river beds (Figure 1B), grab the attention of passers-by, and infuriate anglers by snagging on gear. In some areas, “Didymo” produces a thick, whitish-brown mat composed primarily of excess stalk material that feels like wet cotton and suffocates the river bed. The exact reasons behind the formation of these mats is not known and is a focus of much research. Important macroinvertebrates such as stoneflies, mayflies and caddisflies, the preferred prey of trout species (Marshall et al. 2008), are displaced by the dense mats, and replaced by smaller midges and worms that can easily live in the mat. Because mats tend to occur in cold, clear mountain streams, many recreationists see them as unnatural, ugly, and invasive.

Figure 1. Didymosphenia geminata on stream bottom rock (A) and nuisance mat (B) in the Kootenai River, Libby, MT.
Historically, Didymo is native to the northern latitudes of the world and typically produced mats that lasted only for a few weeks to months (Kilroy 2004, Bothwell and Spaulding 2008). However, in the early 1990’s, nuisance mats (>1 km that persist for several months of the year) began to be recorded in many aquatic systems worldwide. Initially, scientists hypothesized that the species was an invasive, introduced and spread by anglers via contaminated gear, especially felt-soled wading boots. This led to a widespread ban on the use of such boots and the establishment of “Didymo de-contamination” strategies. The invasive species hypothesis was based on macroscopic observations, but the native/invasive nature of the species was not conclusively investigated. The appearance of nuisance mats in the southern hemisphere, notably New Zealand and Chile, however, does suggest some transferability, as there are no historical records of the species at those latitudes.
In Montana’s Kootenai River system, Didymo mats first appeared around 2001, downstream of Libby Dam, where they have remained ubiquitous year-round ever since. Historical reference to the species in fisheries research tells us that it was present in the periphyton pre-impoundment in 1866 (Lord 1866) and post-impoundment (Perry and Huston 1983) but did not occur in mat form. Didymo’s shift from an unseen member of the periphyton community to the cause of prolific nuisance mats has not been fully explained. Currently, the densest mat coverage of Didymo occurs near Libby Dam (up to 30 mm deep with 100% coverage) and continues downriver into Idaho, though with decreasing intensity, forming a gradient from the dam near Libby, Montana. The tail waters of dams have been recognized as “hot spots” for nuisance mats because of low nutrient concentrations, the stable flow regime, and low disturbance to the river bed (Kirkwood et al. 2009).
To understand the distribution of Didymo and its place in the community (mat or regular member of the algae community) in the intermountain Northwest, we sampled 127 streams and rivers in Montana and Idaho (Figure 2) by obtaining scrapings from rocks. We enlisted the help of interested anglers, biologists, and University of Idaho staff and students. Of the 127 sites surveyed, Didymo was present at 65 sites (51%), of which 21 were classified as nuisance mats. At the remaining 44 locations, Didymo was just a regular member of the periphyton. Sixty-two sites did not have any Didymo, which we attributed to the surrounding habitat (high shade, cedar forests and intermittent streams). These data show that Didymo is widespread, and that it usually occurs in a healthy, non-mat phase. This suggests that it is neither invasive nor acting invasively. Our analyses suggest that an interaction between multiple habitat and environmental factors allows the species become a nuisance mat producer.
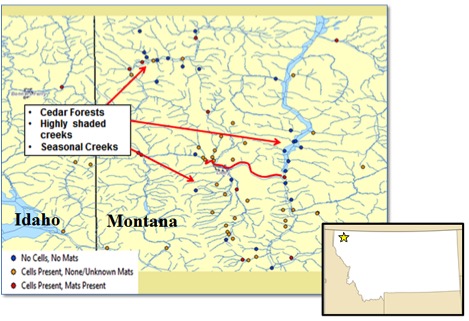
Figure 2. Distribution of Didymosphenia geminata throughout the Kootenai National Forest, Montana, sampled as part of 127 sites in 2014.
To examine this hypothesis, we started controlled experiments in 2013 in a mobile experimental flume system (MEFS) located just below Libby Dam (Figure 3). This system is supplied with water directly from the Kootenai River which is dispersed through 32 independent flumes, each two meters long × 0.2 m wide × 0.05 m tall, and constructed of clear plexiglass. The bottom is covered with replaceable open cell ¼” (6 mm) thick floral foam. In each flume we can independently control the exposure to environmental (e.g., light, water flow, etc.) and chemical (phosphorus and nitrogen) variables to examine the effect of each alone or in combination.

Figure 3. Kootenai River flume study site below Libby Dam in Montana USA. A) Mobile experimental flume systems (MEFS) each with 16 external flumes supplied by water from the Kootenai River. B) Internal structure of MEFS with stock tanks on shelving and mixing buckets below. C) External flumes lined with open celled foam.
Based on previous work of others (Kilroy and Bothwell 2011), we have examined the response of Didymo to various concentrations of dissolved phosphorus for the past two years: 3, 5, and 8 µg/L (March 2013 – September 2013) and 0.5, 1.5, 2, 3, 5 and 8 µg/L (January 2014 – May 2014). Results show that the stalk length (the biomass that forms the nuisance mats) decreased in phosphorus treatments compared to the controls. This suggests that the formation of mats is related to a decrease in the availability of phosphorus.
This makes the onset of mats in the Kootenai River below Libby Dam perplexing, as there were no changes in how phosphorus was managed at the time mats started to form. Recently, Bothwell et al. (2014) suggested that nuisance mat formation occurs not in spite of P limitation but because of it, and that this limitation is mediated by other changes in the environment such as (1) the deposition of reactive nitrogen from the atmosphere, (2) the timing of spring runoff due to climate change, (3) increased nitrogen levels from terrestrial deposition (agriculture and silviculture) and (4) reduced marine-derived nutrients from the decline of anadromous fish populations. These mechanisms would greatly alter the ratio of nitrogen to phosphorus, which may explain the increase in stalk material produced by Didymo. To test this hypothesis, we will alter the addition of nitrogen and phosphorus in the MEFS this coming winter. Our ultimate goal is to identify management strategies by which one could “encourage” Didymo to shift from a nuisance mat to a “well-behaved” (i.e., non-mat forming) member of the algal community to protect the diversity of stream invertebrates and other components of the food web.
Overall, Didymo is perplexing, as it forms nuisance mats under apparently very low nutrient concentrations, while most scientists are used to seeing increased algal biomass with increased nutrients. However, it is this enigma that makes it interesting, and the occurrence of mats may be a sentinel for large-scale ecosystem changes that extend beyond local influences. As history has shown, we would be wise to pay attention to such changes.
References
Bothwell, M.L., and S.A. Spaulding (Co-Editors) 2008. Proceedings of the 2007 International Workshop on Didymosphenia geminata. Can. Tech. Rep. Fish. Aquat. Sci. 2795: xxxv + 58 p.
Bothwell, M.L., B.W. Taylor and C. Kilroy. 2014. The Didymo story: the role of low dissolved phosphorus in the formation of Didymosphenia geminata blooms. Diatom Research. DOI: 10.1080/0269249X.2014.889041.
Kilroy, C. 2004. A new alien diatom, Didymosphenia geminata (Lyngbye) Schmidt – its biology, distribution, effects and potential risks for New Zealand fresh waters: National Institute of Water and Atmospheric Research, New Zealand. Client Report. CHC2004-128.
Kilroy, C. and M.L. Bothwell. 2011. Environmental control of stalk length in the bloom-forming freshwater benthic diatom Didymosphenia geminata (Bacillariophyceae). Journal of Phycology. 47: 981-989.
Kirkwood, A.E., L.J. Jackson and E. McCauley. 2009. Are dams hotspots for Didymosphenia geminata blooms? Freshwater Biology. 54: 1856-1863
Lord J.K. 1866. Naturalist in Vancouver Island and British Columbia. Vols. 1, 2. R. Bentley. Spottiswoode and New-Street Square, London. 375pp.
Perry, S.A. and J.E. Huston. 1983. Section A: Aquatic insect study. October, 1970 – June, 1982. Kootenai River Fisheries Investigations Final Completion Report. pg. 1 – 79.









