By Brianna DeCora, Natural Resources Master’s candidate, Idaho Cooperative Fish and Wildlife Research Unit, University of Idaho, winner of WALPA 2025 Scholarship
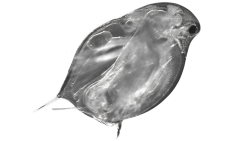
Photo of a female Daphnia pulicaria from American Falls Reservoir, Idaho. The abdominal claw is visible on the left, and an egg is visible in the brood chamber. (Image by Brianna DeCora)
Beneath the surface of Idaho’s lakes and reservoirs, a hidden world of microscopic life is constantly at work. Nestled in the aquatic food chain between algae and fish exist the “animal wanderers” known as zooplankton. Zooplankton serve as trophic intermediaries by consuming algae and transferring energy up the food web to higher trophic levels of planktivorous and piscivorous fish. All fishes rely on zooplankton in some capacity during their life histories, and to fisheries managers, zooplankton are a powerful biotic indicator for food web stability. Understanding variation in zooplankton is critical to understanding the function of lentic systems and guiding fish management actions. Variation in zooplankton density, size structure, and species composition is a primary factor regulating growth rates, survival, and reproductive success of fishes. However, the complexity of zooplankton populations makes gathering representative data on zooplankton populations a challenge to resource managers.
Current methods demand considerable labor, a high degree of taxonomic expertise, time, and considerable resources to obtain accurate data. Abiotic and biotic factors in each waterbody also affect the development of zooplankton sampling techniques. Despite the inherent difficulty of describing their population dynamics, zooplankton serve as a valuable indicator for ecosystem monitoring. The challenges faced in describing zooplankton necessitate the development of rapid assessment techniques that expedite research of trophic interactions. Few zooplankton-based indices and metrics exist that are cost-effective while still producing accurate results in a timely manner. Resource managers for lake systems want a rapid assessment protocol for zooplankton analysis that will help them make informed management decisions.
My master’s work aimed to develop a standardized technique that can be used to rapidly index zooplankton assemblages. The primary focus of my research was to evaluate the effectiveness of sieving zooplankton for use in future lake monitoring efforts.
In the summer of 2024, with the help of Idaho Department of Fish and Game staff, I collected zooplankton from 22 waterbodies across Idaho. In total, I collected 295 zooplankton samples from 64 sites. After sample collection was completed, I processed every sample using both traditional counting techniques and a rapid volumetric method.

The author collecting Zooplankton samples at Arrowrock Reservoir, Boise and Elmore County, Idaho on August 7, 2024. (Image by Matt Corsi)
The rapid method entailed filtering a sample in entirety through a series of seven sieves with mesh sizes ranging from 1000µm to 75µm. Once a sample was thoroughly rinsed through the sieve stack, I carefully transferred the contents of each sieve into a separate graduated cylinder and left the size-fractionated zooplankton to settle for two hours. This method allowed us to determine the biovolume of seven size classes of zooplankton simultaneously. I evaluated the performance of the rapid volumetric method using three criteria: effectiveness, equivalence, and efficiency.
To pair the volumetric data to the traditional density estimates, I collected species-specific measurements and estimated biovolume corrected by abundance. Measurements were also used to confirm which axis was the best predictor for which sieve a given zooplankton would be retained on. The sieves were shown to effectively sort zooplankton by body width (µm; Figure 1). I then calculated proportions of zooplankton size distributions (ZSD) for both the rapid and traditional methods (Figure 2). I also timed how long it took me to process each sample using both methods. I found that, on average, the traditional counting method took 70 minutes, while using the sieve sack to process the same sample took only 26 minutes. Thus, the rapid method reduced processing time by more than 60% (Figure 3).
State agencies need methods that fit real budgets and that provide data in near real time. This rapid sieving method fits budgets, requires no taxonomic expertise, and improves sample processing time by reducing methodological bottlenecks. My goal in completing this thesis is to provide fisheries managers with a tool they can use to bolster their long-term lake monitoring programs. Monitoring zooplankton not only helps show trends in water quality but also provides metrics tied to fish production that can help managers make more informed decisions.
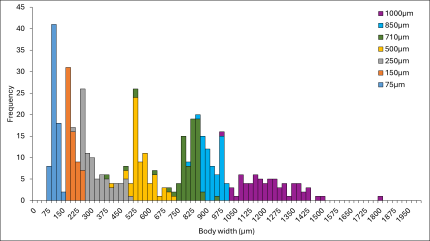
Figure 1. Length-frequency distribution of zooplankton by body width (µm) across seven mesh sizes, showing that body width best predicted passage through each sieve.
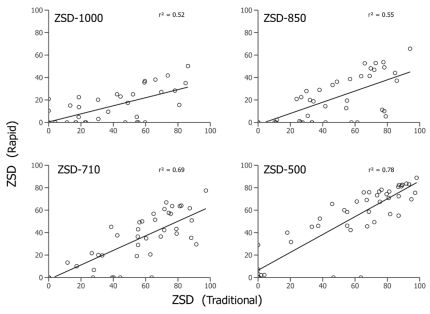
Figure 2. Zooplankton size distribution proportion comparisons for both traditionally estimated biovolume and rapid volumetric estimates, showing highest r2 value (0.78) for ZSD-500.
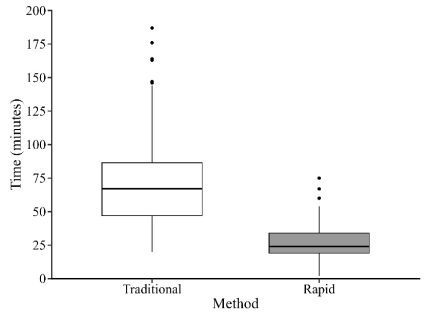
Figure 3. Comparison of processing time showing an average time of 70.4 minutes for traditional methods and 26.1 minutes for the rapid volumetric technique.
Acknowledgements:
I would like to express my heartfelt gratitude to everyone who supported me throughout this project, whether it was by dedicating their time to sampling with me, providing transportation and assistance in sample collection, sharing words of advice, or listening to endless zooplankton facts. Your contributions have been instrumental in advancing this research. I am especially grateful to my advisor Mike Quist for his guidance, encouragement, and unwavering support throughout every stage of this project. This work wouldn’t have been possible without support and funding from the Idaho Department of Fish and Game and the U.S. Geological Survey. Thank you also to Washington State Lake Protection Association for awarding me a scholarship that enabled me to purchase additional graduated cylinders, allowing me to process multiple samples at the same time.


















